UNIT 3 TEST REVIEW
Our test is on Friday, April 11, 2014.
THINGS TO KNOW
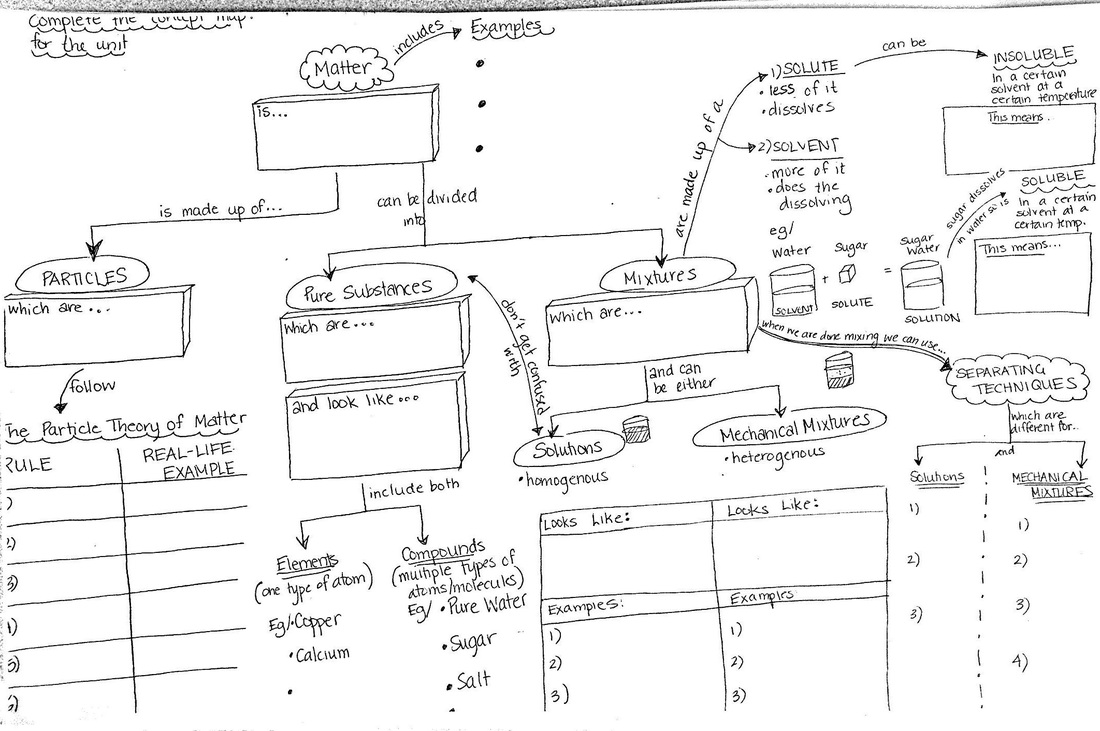
Lesson 1: Matter
Ways of separating solutions (chromatography, distillation, evaporation)
TYPES OF QUESTIONS
• Matching definitions
• Multiple choice
• True/ False, explain
• Diagrams
• Short answer
SAMPLE QUESTIONS
1. Use the terms “dilute” and “concentrated” to explain the difference between frozen juice concentrate and juice.
2. Explain how paper chromatography could be used to separate a mixture of different coloured inks.
3. List three factors that influence the rate at which dissolving occurs.
4. Use the particle theory to explain why hot chocolate powder dissolves more rapidly in hot water than cold water.
5. Explain the difference between an undersaturated, saturated and supersaturated solution.
6. Explain whether you would classify each of the following items as a pure substance or a mixture: apple juice, fruit punch, distilled water, lemonade.
7. Explain why a solid substance occupies less space than the same substance in its gaseous form.
Lesson 1: Matter
- Matter is anything that has mass and takes up space
- Examples of matter
- Homogeneous = SOLUTIONS (same appearance throughout)
- Heterogeneous = MECHANICAL MIXTURES (different appearance throughout)
- The six parts of Particle Theory
- Using Particle Theory to explain experiments
- Solutions are made of a solute (less of it) and a solvent (more of it)
- Solutions can be homogeneous or heterogeneous
- Solutes can be soluble or insoluble in any given solvent at any given temperature
- Solutions can be concentrated or dilute
- Solutions can be undersaturated, saturated and supersaturated
- Temperature
- Particle Size
- Stirring
Ways of separating solutions (chromatography, distillation, evaporation)
TYPES OF QUESTIONS
• Matching definitions
• Multiple choice
• True/ False, explain
• Diagrams
• Short answer
SAMPLE QUESTIONS
1. Use the terms “dilute” and “concentrated” to explain the difference between frozen juice concentrate and juice.
2. Explain how paper chromatography could be used to separate a mixture of different coloured inks.
3. List three factors that influence the rate at which dissolving occurs.
4. Use the particle theory to explain why hot chocolate powder dissolves more rapidly in hot water than cold water.
5. Explain the difference between an undersaturated, saturated and supersaturated solution.
6. Explain whether you would classify each of the following items as a pure substance or a mixture: apple juice, fruit punch, distilled water, lemonade.
7. Explain why a solid substance occupies less space than the same substance in its gaseous form.
| science_unit_3_test_review.pdf | |
| File Size: | 242 kb |
| File Type: | |
| unit_3_concept_map.pdf | |
| File Size: | 376 kb |
| File Type: | |